Research Grade Blisibimod
All
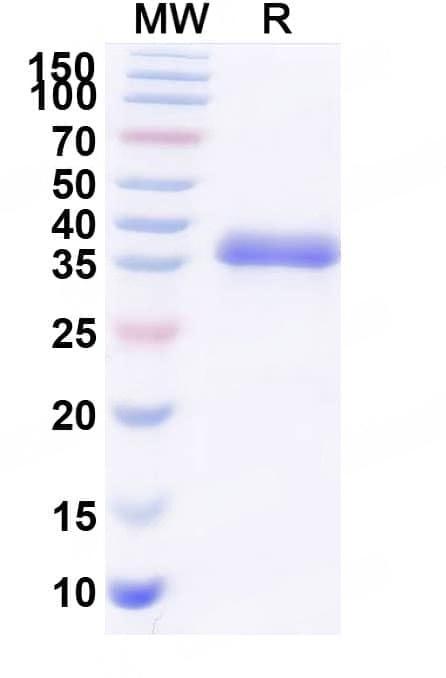
SDS-PAGE

- Catalog: TD-HV212056
- Clonality:
- Application: ELISA, Functional assay, Research in vivo
- Synonyms: A-623
-
规格:
询价
-
价格:
¥100ug
-
Catalog No.TD-HV212056
-
DescriptionBlisibimod(TD-HV212056) is a research-grade recombinant antibody targetingCD257. Produced in mammalian cells with native-like glycosylation.HighlightsResearch Grade— For PK/PD studies, assay development, and ADA research.Native Glycosylation— Mammalian expression ensures native-like patterns.
-
Species reactivityHuman
-
ApplicationsELISA, Functional assay, Research in vivo
-
Host speciesHuman
-
IsotypeFusion - [peptide 16-mer - peptide 19-mer]2 - IGHG1 Fc (Fragment constant)
-
TargetBAFF, ZTNF4, BLYS, Tumor necrosis factor ligand superfamily member 13B, Dendritic cell-derived TNF-like molecule, TALL-1, TNF- and APOL-related leukocyte expressed ligand 1, BLyS, B lymphocyte stimulator, TNFSF13B, CD257, TNFSF20, TALL1, B-cell-activating factor
-
Endotoxin levelPlease contact the lab for this information.
-
Purity>95% purity as determined by SDS-PAGE.
-
PurificationProtein A/G purified from cell culture supernatant.
-
AccessionQ9Y275
-
FormLiquid
-
Storage buffer0.01M PBS, pH 7.4.Please refer to the specific buffer information in the hardcopy of datasheet or the lot-specific COA.
-
Stability and StorageUse a manual defrost freezer and avoid repeated freeze-thaw cycles. Store at 4°C for short-term storage (1-2 weeks). Store at -20°C for up to 12 months. For long-term storage, store at -80°C.
-
Alternate NamesA-623
-
BackgroundBlisibimod (also known as A-623, formerly AMG 623) is a selective antagonist of B-cell activating factor (BAFF, also known as B-lymphocyte stimulator or BLyS), being developed by Anthera Pharmaceuticals as a treatment for systemic lupus erythematosus. It is currently under active investigation in clinical trials.Mechanism of actionBlisibimod is a fusion protein consisting of four BAFF binding domains fused to the N-terminus of the fragment crystallizable region (Fc) of a human antibody.BAFF is involved in B-cell survival, activation, and differentiation. Elevated levels of BAFF have been associated with several B-cell mediated autoimmune diseases, including systemic lupus erythematosus, lupus nephritis, rheumatoid arthritis, multiple sclerosis, Sjögren syndrome, Graves' disease, and Hashimoto's thyroiditis. Blisibimod binds to BAFF and inhibits interaction with BAFF receptors, thus decreasing B-cell survival and proliferation throughout the body. Improvements in disease activity have been observed in patients with systemic lupus erythematosus and rheumatoid arthritis following treatment with BAFF inhibitors in clinical trials.DevelopmentBlisibimod was initially developed by Amgen, with Phase I trials demonstrating comparable safety between the blisibimod and placebo treatments. It was subsequently acquired by Anthera Pharmaceuticals, who in 2010 initiated a global Phase II study called PEARL-SC to investigate the efficacy, safety, and tolerability of blisibimod in subjects with systemic lupus erythematosus. The PEARL-SC study, completed in April 2012, yielded data that has been published. Blisibimod is currently being tested in a Phase 3 study, CHABLIS-SC1, for systemic lupus erythematosus, and a Phase 2 study, BRIGHT-SC, for IgA nephropathy.• Pharmacokinetics and immunological effects of exogenously administered recombinant human B lymphocyte stimulator (BLyS) in mice., PMID:11160623
-
CaptionSDS-PAGE for Research Grade Blisibimod
-
NoteFor research use only. Not suitable for clinical or therapeutic use.
