Research Grade Idarucizumab
All
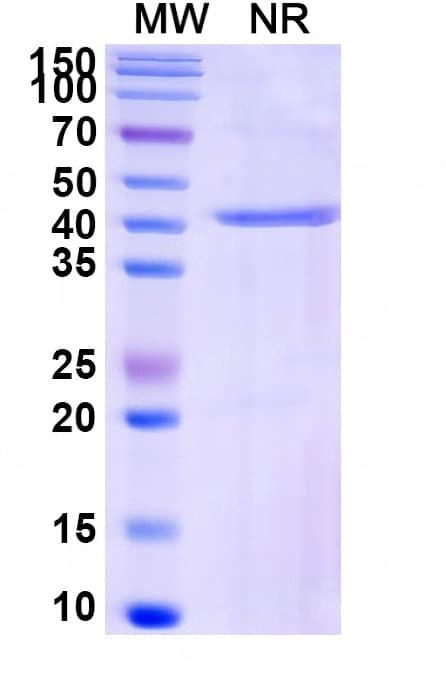
SDS-PAGE

- Catalog: TD-YP598016
- Clonality: Monoclonal
- Application: ELISA, Bioactivity: FACS, Functional assay, Research in vivo
- Synonyms: aDabi-Fab, anti-dabigatran, antidotefordabigatran, CAS: 1362509-93-0
-
规格:
询价
-
价格:
¥100ug
-
Catalog No.TD-YP598016
-
DescriptionIdarucizumab(TD-YP598016) is a research-grade recombinant antibody targetingDabigatran Etexilate Mesylate. Produced in mammalian cells with native-like glycosylation.HighlightsResearch Grade— For PK/PD studies, assay development, and ADA research.Native Glycosylation— Mammalian expression ensures native-like patterns.
-
Species reactivityGeneral
-
ApplicationsELISA, Bioactivity: FACS, Functional assay, Research in vivo
-
Host speciesHumanized
-
IsotypeFab-G1-kappa
-
ClonalityMonoclonal
-
TargetDabigatran Etexilate Mesylate
-
Endotoxin levelPlease contact the lab for this information.
-
Purity>95% purity as determined by SDS-PAGE.
-
PurificationProtein A/G purified from cell culture supernatant.
-
Accession211915-06-9
-
FormLiquid
-
Storage buffer0.01M PBS, pH 7.4.Please refer to the specific buffer information in the hardcopy of datasheet or the lot-specific COA.
-
Stability and StorageUse a manual defrost freezer and avoid repeated freeze-thaw cycles. Store at 4°C for short-term storage (1-2 weeks). Store at -20°C for up to 12 months. For long-term storage, store at -80°C.
-
Alternate NamesaDabi-Fab, anti-dabigatran, antidotefordabigatran, CAS: 1362509-93-0
-
BackgroundIdarucizumab is a humanized monoclonal antibody fragment. Idarucizumab is first reversal agent for a direct oral anticoagulant (DOAC). Idarucizumab can specifically neutralize the effects of the oral direct thrombin inhibitor in order to restore hemostasis.• Dabigatran etexilate for the treatment of acute venous thromboembolism in children (DIVERSITY): a randomised, controlled, open-label, phase 2b/3, non-inferiority trial., PMID:33290737• Dabigatran Etexilate: A Review in Pediatric Venous Thromboembolism., PMID:35699915• The polymorph landscape of dabigatran etexilate mesylate: Taking the challenge to bring a metastable polymorph to market., PMID:37044200• Dose tailoring of dabigatran etexilate: obvious or excessive?, PMID:25994994• A Pharmacokinetic Evaluation of Dabigatran Etexilate, Total Dabigatran, and Unconjugated Dabigatran Following the Administration of Dabigatran Etexilate Mesylate Capsules in Healthy Male and Female Subjects., PMID:31652461• Idarucizumab for Dabigatran Reversal - Full Cohort Analysis., PMID:28693366• Dabigatran Etexilate: A Review in Nonvalvular Atrial Fibrillation., PMID:28185082• Direct oral anticoagulants: evidence and unresolved issues., PMID:33248499• Transfer of dabigatran and dabigatran etexilate mesylate across the dually perfused human placenta., PMID:24807346• The discovery of dabigatran etexilate for the treatment of venous thrombosis., PMID:27159158
-
CaptionSDS-PAGE for Research Grade Idarucizumab
-
NoteFor research use only. Not suitable for clinical or therapeutic use.
